A pH Analyser is a vital tool in various industries. It measures the acidity or alkalinity of solutions, providing essential data for quality control. According to recent market research, the global pH meter market is projected to grow significantly, estimated at over $1 billion by 2026. This underscores the importance of accurate pH measurement in sectors such as food and beverage, pharmaceuticals, and water treatment.
Understanding how a pH Analyser works is crucial for effective application. These instruments utilize glass electrodes that respond to hydrogen ion concentration, delivering precise readings. However, calibration is often overlooked, leading to inaccurate results. Inaccuracies can cause costly errors in product quality, affecting consumer trust and brand reputation.
Moreover, using a pH Analyser is not without challenges. Environmental factors, such as temperature and contamination, can interfere with measurements. Regular maintenance and proper handling are essential to ensure reliability. Despite advancements, many industries still face issues that warrant reflection on existing practices. Investing in training and technology can enhance the effectiveness of pH analysers in any operation.

A pH analyser is a device used to measure the acidity or alkalinity of a solution. It provides a numerical value representing the pH level. This level indicates how acidic or basic a substance is, which is crucial for many industries. For example, agriculture, water quality testing, and food production all rely on precise pH measurements.
Using a pH analyser involves inserting a probe into the solution. The probe converts the pH level into an electrical signal. This signal is then displayed on a screen. Calibration is necessary for accurate results. Ensure you use fresh calibration solutions to avoid errors.
Tips: Always rinse the probe with distilled water before use. This helps to prevent contamination. Keep the probe clean and stored correctly. A dirty probe can lead to inaccurate readings. Consider recording the pH levels regularly. This practice can help identify trends over time.
The history and development of pH analysers date back to the early 1900s. The
Glass Electrode, a significant breakthrough, was invented in 1934 by the chemist A. Harned.
This innovation allowed for accurate measurements of hydrogen ion concentration in various solutions. The need for precision in
industries like agriculture, food science, and pharmaceuticals amplified the demand
for robust pH measurement tools.
By the 1950s, pH meters became more accessible. The technology evolved rapidly with the introduction of solid-state electronics.
According to a 2021 industry report, the global pH meter market size reached $450 million.
This growth is attributed to increased agricultural practices and stringent regulations in water quality monitoring.
Knowledge of pH levels is integral in ensuring safety and quality across many sectors.
Tips: Regular calibration is essential for maintaining accuracy.
Even a slight deviation can lead to significant errors in results. It's wise to perform checks daily, especially in
critical processes. Monitoring the pH can sometimes be challenging. Factors like temperature and ionic strength can affect readings.
Users should understand these variables to mitigate potential errors.
pH analysers play a crucial role in measuring acidity in various industries. They operate on the principle of ion exchange and potential measurement. The basic design includes a glass electrode, which is sensitive to hydrogen ions. When immersed in a solution, the electrode generates a voltage that is proportional to the hydrogen ion concentration.
According to a report from the International Society for Measurement and Control, proper calibration of pH analysers is essential. A drift in calibration can lead to inaccuracies exceeding 0.5 pH units. Regular maintenance and routine checks are necessary. Many industries experience challenges in achieving consistent results. Factors like temperature and solution composition can affect readings.
The latest advancements in technology include digital pH sensors. These sensors offer improved accuracy and faster response times. However, they require specific operating conditions to function optimally. In many cases, users still report occasional discrepancies. These actions prompt regular training for staff to understand the intricacies of pH measurement. Adjustments in testing protocols often reveal the need for scrutiny in routine practices. Ensuring reliable pH readings remains a continuous effort for many professionals in the field.
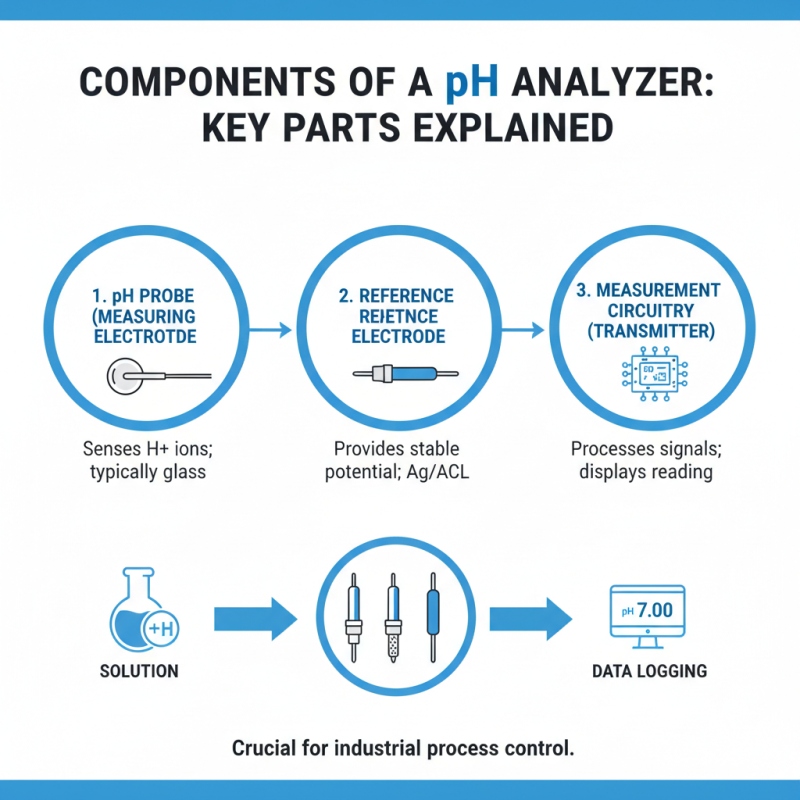
A pH analyser is a crucial instrument in various industries, designed to measure the acidity or alkalinity of a solution. Understanding its components is essential for effective operation and maintenance. The key parts include the pH probe, the reference electrode, and the measurement circuitry.
The pH probe is the most vital component. It contains a glass membrane that responds to hydrogen ion concentration. This membrane can degrade over time, leading to inaccurate readings. Regular calibration and cleaning are necessary to maintain accuracy. According to a recent industry report, over 30% of pH probes fail due to neglect and improper handling.
The reference electrode stabilizes the measurement, providing a known voltage. Without it, readings can be unreliable. Measurement circuitry converts the probe's signals into readable values. It's worth noting that these circuits are sensitive to electrical interference. As reported, nearly 25% of pH analyser malfunctions in industrial settings stem from faulty circuitry. Routine checks and adjustments could prevent these issues. Understanding these components ensures better performance and longevity of pH analysers.
pH analysers play a critical role across numerous industries. In environmental monitoring, they help ensure water quality. According to a report by the Environmental Protection Agency, pH levels in fresh water must remain between 6.5 to 8.5 for aquatic life. A slight deviation can lead to severe ecological impacts. Continuous pH monitoring helps avoid these risks.
In the food and beverage sector, pH control is vital. Fermentation processes depend on precise pH levels. For example, a study highlighted that optimal pH for yogurt production is around 4.6. Incorrect pH can lead to spoilage and impacting product safety. This is especially important as consumer demand grows for high-quality, safe products.
Pharmaceutical companies utilize pH analysers to ensure drug efficacy. The stability of many active ingredients is affected by pH. Research published in the Journal of Pharmaceutical Sciences indicates that 75% of drug formulations require strict pH control. Failing to monitor pH can result in ineffective medications. Industries must continuously improve their pH monitoring practices to maintain quality.
| Industry | Application | Importance of pH Analysis | Typical pH Range |
|---|---|---|---|
| Food & Beverage | Quality Control | Ensures safety and taste | 3.5 - 6.5 |
| Agriculture | Soil Testing | Optimizes crop yield | 6.0 - 7.5 |
| Wastewater Treatment | Monitoring Effluents | Protects ecosystems | 6.5 - 9.0 |
| Pharmaceuticals | Product Formulation | Ensures efficacy and safety | 4.0 - 7.0 |
| Environmental Monitoring | Water Quality Assessment | Tracks pollution levels | 6.0 - 8.5 |