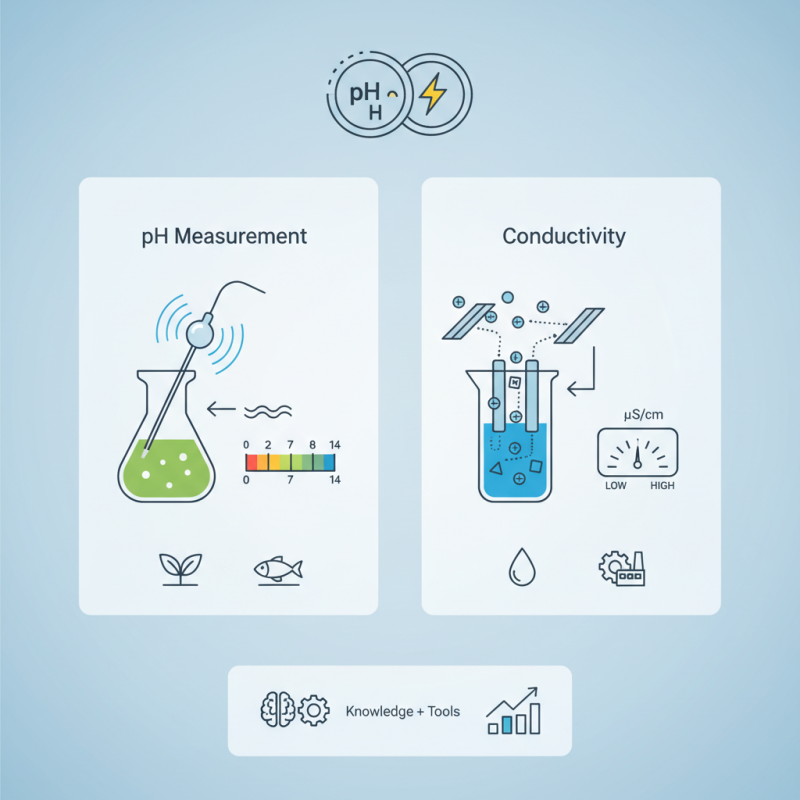
The pH conductivity meter is a vital tool in various industries, including agriculture, environmental monitoring, and food production. According to Dr. Sarah Thompson, a leading expert in analytical chemistry, "Understanding the parameters of pH and conductivity is crucial for maintaining quality in any water-based solution." This instrument not only measures pH levels but also the conductivity of a solution, offering insights into its ionic content.
A pH conductivity meter functions by employing electrodes that respond to ion concentrations in a solution. It translates these interactions into readable data. This information can be pivotal for scientists and engineers alike. For instance, the meter can help determine the health of aquatic ecosystems or monitor nutrient solutions in hydroponics. However, not all pH conductivity meters are created equal. Some may lack accuracy, making it essential for users to evaluate their options carefully.
In today's advancing technological landscape, the importance of quality measurement tools is undeniable. The reliance on pH conductivity meters reflects a growing need for precision. Professionals must be aware of the limitations and potential errors associated with their use. The device, while beneficial, requires an understanding of how to interpret its data properly. This balance between tools and knowledge is what drives success in this field.
A pH conductivity meter is a vital tool used in various fields such as agriculture, environmental science, and laboratory research. It measures the acidity or alkalinity of a solution by determining its pH level while also gauging electrical conductivity. This dual functionality allows users to assess water quality and nutrient levels in one device. Understanding how it works can greatly enhance the accuracy of scientific experiments.
The pH conductivity meter operates by using electrodes that sense the concentration of hydrogen ions in a solution. These electrodes generate a small voltage that corresponds to the pH reading. Meanwhile, conductivity is measured by the solution's ability to conduct electricity, indicating the presence of dissolved ions. It’s fascinating how something so compact can provide critical information about water quality. However, it can be tricky; calibration and maintenance are essential for reliable results. Neglecting these steps might lead to inaccurate readings and flawed interpretations.
For those venturing into using a pH conductivity meter, experience plays a significant role. Proper training is vital. A user must learn to interpret the data accurately. There are challenges, like interference from temperature and other chemical substances. Being aware of these issues can prevent misleading conclusions. A pH conductivity meter is powerful, yet it demands respect and careful handling for optimal results.
A pH conductivity meter is essential for measuring the acidity or alkalinity of a solution. Understanding its key components can enhance its effective use in laboratories or fieldwork.
The most crucial part of the pH meter is the electrode. This glass component senses hydrogen ion concentration. It generates a voltage that correlates with the solution's pH. Proper calibration of the electrode is vital. If not calibrated correctly, readings can be inaccurate. Regular maintenance is needed to prolong its life.
Another key component is the temperature sensor. Temperature affects pH readings. A good meter compensates for temperature, providing accurate results across various conditions. Additionally, the display interface is important. It should show real-time data clearly. Some meters include features like data logging. This can improve efficiency, but reliance on technology may sometimes result in errors if overlooked.
A pH conductivity meter is essential in monitoring water quality and chemical processes. Its operation is grounded in two fundamental principles: measuring pH levels and assessing electrical conductivity. The pH measurement relies on the hydrogen ion concentration in a solution. Accurate sensors detect these ions and convert the data into an understandable format.
In terms of conductivity, this refers to a solution's ability to conduct electric current. The more ions present, the higher the conductivity. According to recent industry reports, sodium chloride solutions can have a conductivity range of 0 to 100,000 μS/cm. This spectrum is critical for various applications, including environmental monitoring, agriculture, and industrial processes. However, several factors influence these measurements, such as temperature and ionic strength.
It’s important to identify calibration needs to ensure accuracy. The calibration process can be challenging. Users must consider the purity of calibration solutions, and any contaminants may compromise results. An experiment revealed that a well-maintained meter can yield results within 0.01 pH units, but operational errors can lead to inconsistencies. Understanding these operational principles allows for more precise control over the processes where these measurements are utilized.
| Measurement Parameter | Unit | Typical Range | Application |
|---|---|---|---|
| pH | pH units | 0 - 14 | Water quality testing |
| Conductivity | µS/cm | 0 - 20000 | Nutrient solution monitoring |
| Temperature | °C | 0 - 100 | Laboratory experiments |
| Salinity | ppt | 0 - 35 | Aquatic ecosystem studies |
pH conductivity meters play a vital role in various industries. They measure the pH level and electrical conductivity of solutions. This data is crucial for many applications.
In agriculture, these meters are used to monitor soil health. A study by the Agriculture Department highlights that optimal pH levels can enhance crop yield by up to 30%. Farmers can adjust soil conditions based on meter readings, ensuring better growth and sustainable practices.
In water treatment, pH conductivity meters help maintain water quality. The Water Research Foundation indicates that proper pH balance reduces the risk of bacterial growth. Facilities can operate more efficiently by frequently checking these parameters.
Tips: Regular calibration of pH meters is essential for accurate readings. Keep your device clean to extend its lifespan. Periodic maintenance helps ensure reliable performance over time.

pH conductivity meters are vital tools in various fields, including environmental monitoring and agriculture. To ensure accuracy, regular maintenance and calibration are essential. According to a report from the American Society for Testing and Materials (ASTM), improper calibration can lead to measurement errors exceeding 10%. This highlights the importance of routine checks.
Calibration should be performed using standardized buffer solutions. These solutions must closely match the expected pH range of the samples. Maintaining these standards in proper conditions helps prevent contamination and degradation. Calibration should be scheduled monthly, or more frequently if readings fluctuate significantly.
Tips: Always document calibration dates to track your meter’s performance. Consider investing in a calibration kit that includes multiple pH buffers. This often yields better results. Regular cleaning of the electrode is crucial too. Any residue can skew results, ultimately affecting your data integrity. Monitoring your equipment will help identify issues early and save time on repairs.