Accurate measurements in various industries rely heavily on the precision of tools like the pH conductivity meter. This instrument measures both the pH level and the electrical conductivity of fluids, providing crucial data for laboratory and field applications. According to a recent report by the Global pH Meter Market, the demand for reliable pH monitoring is projected to grow by 6.5% annually over the next five years.
Dr. Emily Chen, an expert in analytical chemistry, emphasizes this importance: “The accuracy of a pH conductivity meter can significantly impact research outcomes.” Her insight highlights the role of this device in agriculture, environmental science, and pharmaceuticals, where precision is paramount.
However, using a pH conductivity meter requires more than just technical knowledge. Users often face challenges in calibration and maintenance, which can lead to misleading results. Understanding these limitations is essential for obtaining reliable data. Proper knowledge and training can enhance the effectiveness of a pH conductivity meter, ensuring that measurements are both dependable and informative.
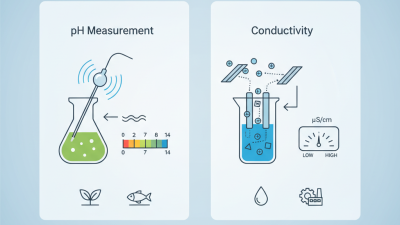
pH conductivity meters are essential tools in various scientific and industrial applications. They measure the acidity or alkalinity of a solution, which is crucial in fields like agriculture, environmental science, and water treatment. These meters work by sensing the hydrogen ion concentration, providing precise readings that aid in maintaining optimal conditions for various processes.
Understanding how these meters function is key to using them effectively. The device typically has two electrodes that measure electrical conductivity as a means to estimate pH. Factors like temperature and ionic strength can affect accuracy. Regular calibration is vital, as it helps maintain reliability. Users often overlook this step. Neglecting calibration can lead to significant errors in measurement.
An essential aspect of using pH conductivity meters is proper electrode care. Rinsing the electrode with distilled water before and after use is often forgotten. This simple task can prevent contamination and improve accuracy. Furthermore, data interpretation may seem straightforward, but it can be complex. Understanding local water chemistry and environmental factors is crucial for making accurate assessments.
Preparing a pH conductivity meter for accurate measurements requires careful attention. Start by ensuring the meter is clean and properly calibrated. Use a standard buffer solution to calibrate the pH readings. Immerse the probe in the solution and adjust the meter until it reads the buffer's pH accurately. This step is crucial, yet often overlooked.
Next, inspect the electrode for any signs of damage or contamination. A dirty probe can lead to inconsistent readings. Rinse it with distilled water before testing. This simple action can enhance reliability significantly. After calibration and cleaning, hydrate the sensor in a pH storage solution if necessary. This step maintains the probe’s longevity.
While these steps seem simple, many users overlook them. Neglecting calibration or using a dirty electrode can skew results. Taking time to prepare the equipment ensures more accurate readings and reliable data. Such attention to detail fosters better results in experiments.
| Measurement Parameter | Unit | Typical Value | Importance |
|---|---|---|---|
| pH Level | pH | 6.5 - 7.5 | Indicates acidity/alkalinity |
| Conductivity | μS/cm | 200 - 800 | Measures ionic concentration |
| Temperature | °C | 20 - 25 | Affects pH and conductivity |
| TDS (Total Dissolved Solids) | mg/L | 100 - 500 | Reflects water quality |
Calibrating a pH conductivity meter is crucial for obtaining reliable measurements. A study published by the American Society for Testing and Materials highlights that accuracy in pH measurement affects not only experimental results but also product quality in various industries. To begin, ensure your calibration solutions are fresh and appropriate, typically at pH 4, 7, and 10. Measurements can drift over time, so checking before each use is essential.
Carefully rinse the electrode with distilled water to avoid contamination. Immerse the electrode in the first calibration solution and allow it to stabilize, which can take several minutes. Note that temperature can impact readings, so using a temperature-compensated meter can be beneficial. The ideal calibration process should be conducted at room temperature, around 25°C, as variations can lead to a 0.01 pH shift, according to the Journal of Chemical Education.
After calibration, test the meter with another standard solution to ensure accuracy. If there are discrepancies, re-calibrate. Regular maintenance of the electrode is often overlooked. Pollutants can build up, affecting performance. If you notice inconsistency after calibration, it might be time to clean or replace the electrode. Keeping records of calibration dates and results enhances reliability in your measurements.

When measuring pH and conductivity accurately, it’s essential to prepare your equipment properly. Ensure your pH conductivity meter is calibrated. Calibration should match the specific range of your samples, which increases measurement reliability. Be mindful of the temperature, as pH readings can fluctuate with temperature changes.
Tips: Always rinse the electrode with distilled water before and after taking measurements. This practice prevents contamination and improves accuracy. Keep air bubbles away from the electrode; they can lead to misleading results.
When taking readings, immerse the electrode fully in the sample. If the electrode isn’t completely submerged, the readings could be skewed. Allow adequate stabilization time for the meter to ensure accurate results. Not all samples respond the same; some may require multiple readings to find an average.
Tips: If your readings fluctuate, try ensuring the sample is well mixed. Uneven distribution can affect conductivity. Remember, the surface of your samples should be clean. Sediments or residue can lead to issues during measurement.
Accurate pH measurements require careful interpretation and recording of results. Start by calibrating your pH conductivity meter regularly. This ensures precision in your readings. When testing samples, make sure to stir them gently to allow for an even distribution. A stable reading appears on the display when the electrode is adequately submerged.
Interpretation can be tricky. Watch out for fluctuating readings, as these may indicate interference. Consider factors such as temperature and ionic strength, which can impact accuracy. Note any anomalies during testing. Record all measurements immediately, including time and sample conditions, to maintain a reliable dataset.
Tips: Maintain a consistent testing environment to reduce variability. Clean the electrode after each test to prevent contamination. Use a notebook or digital device to keep track of your observations, enhancing clarity and reducing errors in data collection. Review your recorded data periodically to spot trends or irregularities.